What Do Lortab Pills Look Like?

Written by: Fikret Terzic MD, MS
Lortab may take the form of oval tablets colored white, white with colored specials, or pink. Solid colored generic forms of the drug and a liquid Lortab formulation are also available
Lortab is a brand name prescription painkiller product used to treat moderate to severe pain. While liquid Lortab formulations are still available for medical use, Lortab pills have been discontinued.
What Do Lortab Pills Look Like?
Lortab pills contain varying doses of hydrocodone and are distinguished with different tablet colors and designs.
These strengths included:
5 mg hydrocodone / 325 acetaminophen: an oval, bisected, white tablet imprinted with “G 035”

7.5 mg / 325: an oval, bisected, white tablet imprinted with “G 036”

10 mg / 325: an oval, bisected, white tablet imprinted with “G 037”

5 mg / 500: an oval, bisected white tablet with pink speckles, imprinted with “ucb 901”
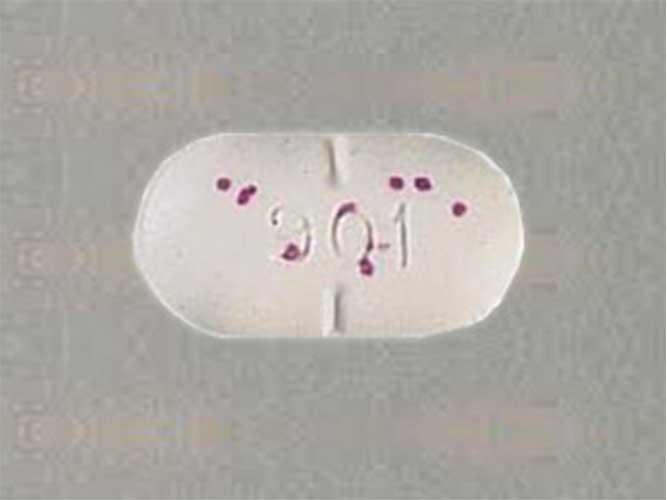
5 mg / 500: an oval, bisected white tablet with blue speckles, imprinted with “ucb 902”
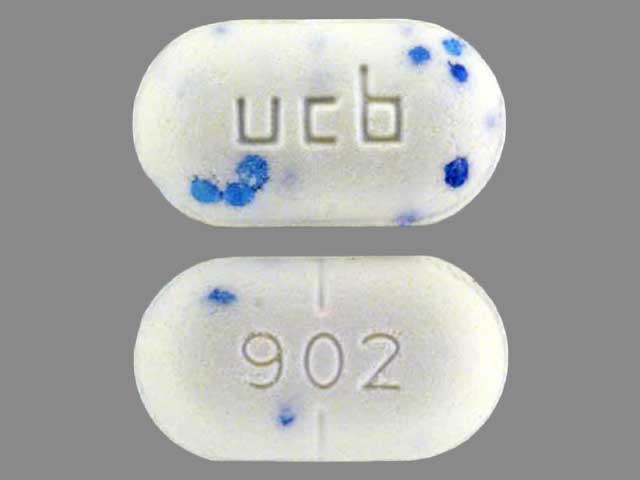
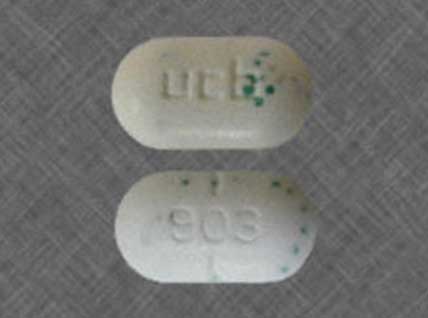
7.5 mg / 500: an oval, bisected white tablet with green speckles, imprinted with “ucb 903”
10 mg / 500: an oval, bisected pink tablet, imprinted with “ucb 910”

Blue & Yellow Lortab
Note that blue Lortab (also discontinued) and yellow Lortab likely refer to generic versions of Lortab 10/500 and 10/325, respectively, as these generic pain medications come in solid colors and are effectively equivalent to the brand name products.
Fake Lortab Pills
Due to high demand for opioid drugs outside of the medical system, many prescription medications are counterfeited in overseas drug labs.
This includes Lortab, which may be faked using any number of unregulated ingredients including fentanyl, methamphetamine, and ketamine.
Counterfeit opioid use is extremely hazardous, often leading to serious side effects including opioid overdose, shallow breathing, and respiratory depression. However, these life-threatening side effects can be reversed with an immediate dose of naloxone.
Acetaminophen Limits & Lortab Discontinuation
As of 2014 the FDA has limited the dosage for acetaminophen combination drugs to 325 mg. This was due to a lack of evidence that higher doses of the analgesic provided any greater pain relief, and urgent concerns over liver damage stemming from high-dose acetaminophen use.
As a result, all Lortab versions containing 500 mg of acetaminophen were discontinued in 2014, before the remainder of the brand was taken off the market in the years that followed.
Generic forms of hydrocodone and acetaminophen are still often referred to as Lortab in common use.
About Lortab
Lortab is a combination opioid medication made with both hydrocodone, an opioid/opiate narcotic that stimulates one’s opioid receptors in the central nervous system, and the over-the-counter pain reliever acetaminophen, also sold as Tylenol.
As with other prescription drugs that contain opioids, Lortab is classified as a Schedule II controlled substance with a high potential for abuse, dependence, and addiction.
Similar combination painkiller medications include:
- Norco (hydrocodone bitartrate and acetaminophen)
- Vicodin (hydrocodone bitartrate and acetaminophen)
- Lorcet (hydrocodone bitartrate and acetaminophen)
- Percocet (oxycodone and acetaminophen)
Lortab Side Effects
The most common side effects reported with the use of acetaminophen/hydrocodone products may include:
- anxiety
- blurred vision
- constipation
- disorganized thoughts
- dizziness
- drowsiness
- dry mouth
- headache
- lightheadedness
- mood changes
- nausea
- ringing in your ears
- upset stomach
- vomiting
However, other uncommon and sometimes serious side-effects and adverse reactions have also been reported, including allergic reactions.
Lortab Drug Interactions
Lortab may interact with other drugs and supplements including:
- other products containing acetaminophen, due to the risk of liver toxicity
- other CNS depressants including alcohol, sedatives, muscle relaxers, and others, due to the risk of respiratory depression
- MAO inhibitors, as a life-threatening reaction may occur
- grapefruit juice, which can increase the drug’s potency
Other Precautions & Warnings
According to information published by the FDA, Lortab and similar medications may also cause:
- sleep-related breathing problems
- adrenal gland problems
- harmful effects relating to pregnancy and breast milk/breastfeeding
- serious skin reactions
The drug should be used with caution or avoided by those with medical conditions including low blood pressure, adrenal gland problems, a history of drug or alcohol abuse, breathing problems, head injuries, kidney or liver disease, and other conditions.
Prolonged use of the drug may also lead to physical dependence, tolerance, and withdrawal symptoms when Lortab is discontinued.
Lortab Abuse & Addiction
Like other opioid pain medications, Lortab and similar medications are diverted for drug abuse. Taken in higher doses (despite the risk of overdose and acetaminophen poisoning), Lortab can provide an addictive, habit-forming drug high.
Signs of Lortab abuse and addiction can include:
- using the drug when it was not prescribed to you
- taking more of the drug or taking it more often than prescribed
- going out of your way to get more of the drug, often through theft, illicit purchases, or doctor shopping
- mood changes
- spending excess time or money thinking about, obtaining, or using the drug
For information on our inpatient treatment options for opioid addiction, please contact Ohio Recovery Center today.
- Food and Drug Administration (FDA) — All manufacturers of prescription combination drug products with more than 325 mg of acetaminophen have discontinued marketing https://www.fda.gov/drugs/information-drug-class/all-manufacturers-prescription-combination-drug-products-more-325-mg-acetaminophen-have-discontinued
- Food and Drug Administration (FDA) — NORCO® Hydrocodone Bitartrate and Acetaminophen Tablets, USP 5 mg/325 mg CII https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/040099s023lbl.pdf
- National Library of Medicine: MedlinePlus — Hydrocodone Combination Products https://medlineplus.gov/druginfo/meds/a601006.html